What is Traceability?
The word traceability is used in many industries, from food to the systems and software development sector and even in forestry. The National Institute for Standards and Technology (NIST) uses the words “documented unbroken chain” to describe traceability.
In every use of traceability, the beginning origin must be known for the product or system to be traceable. The same is true for the serum industry. With serum, traceability encompasses the ability to follow unbroken documentation through every step in the process starting with where the blood was collected. This is the origin as stated on product labels. This process is a core component of the ISIA ethos. What you see on the label must be a true and accurate description of what is in the bottle.
_________________________________________________________________________________________________________________________________________________________________________________________________
Traceability Certification
ISIA traceability certification is based on an independent third-party audit. These audits are only conducted by ISIA approved and trained auditors with an approved audit plan. Any nonconformities identified are categorized and reported to the auditee in a timely manner.
Traceability certification is not available for purchase, nor can it be transferred under any circumstances. Purchase of material from a certified company, or processing of material by a certified company, confers no right to claim or imply traceability certification.
Initial certification requires an audit of at least one year of data and is granted for three years. Recertification audits review the last three years of data. ISIA will not comment on claims made prior to the formation of the Association.
Each audit covers all aspects of the product, at every step - from the abattoir to the end user, including all transactions within the company. Ordering and shipment details for companies who are distributors may be included. While it is not a financial audit, review of invoices and other financial records are essential to determine quantities of material moved and evidence that payments have been made.
Companies are certified for the piece of the process they perform. This may be one step or, for vertically integrated companies, it may be every step.
_________________________________________________________________________________________________________________________________________________________________________________________________
Origin
The ISIA has developed definitions of origin, based on international guidelines set by the WTO, OIE, as well as the USDA and the EU. Since the word “origin” has several meanings and can cause confusion and controversy, the ISIA suggests the use of very precise wording whenever possible.
- Country of Collection - This is the country where the animal was slaughtered and the blood was collected or, in the case of donor animals, where the blood was collected. This includes animals born and raised in the country of collection, as well as imported animals, and may include animals imported for immediate slaughter. This is the country shown on the label as “origin”. The Country of Collection is required to be shown on the Certificate of Analysis (CoA) and Certificate of Origin (CoO). The process of collecting blood and separating serum is considered substantial transformation.
- Country of Manufacturing - This is the country where filtration, bottling, and labeling occur and includes any other processes performed prior to filtration and bottling. Other acceptable terms include “Manufacturing Address” or “Site of Manufacture”. This should be listed on the CoA. This is not considered substantial transformation.
- Country of Further Treatment – This is the country in which the product is treated after sterile filtration. Examples of further treatment are gamma irradiation or heat inactivation. These processes should be listed individually on the CoA and are not considered substantial transformation.
- The Country of Collection (“origin” on the label) does NOT change for import/export purposes. The Country of Collection can never change.
Click here to view the official ISIA Origin Definitions document
_________________________________________________________________________________________________________________________________________________________________________________________________
For a list of ISIA Traceability Certified Companies, Click here
Detailed information regarding the ISIA Traceability Certification Program, Click here
_________________________________________________________________________________________________________________________________________________________________________________________________
Dr. Versteegen, Subject Matter Expert (former CEO), discusses the benefits of being ISIA Traceability Certified.
_________________________________________________________________________________________________________________________________________________________________________________________________
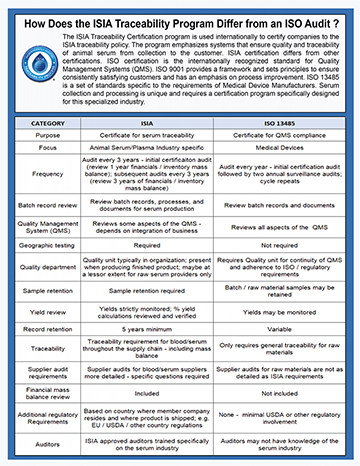
How Does the ISO Audit Differ from the ISIA Traceability Program?
Click here to view and/or download this important info you can review and share with customers.
_________________________________________________________________________________________________________________________________________________________________________________________________
Get the confidence that your serum is manufactured under the highest traceability standards. Make sure you have the transparency and confidence you deserve as nothing works like serum.
Click here to learn why more and more researchers look for the ISIA Traceability certification seal before they select their serum supplier.


